Gambo rides again: the beaked beast of Bungalow Beach and the prehistoric survivor paradigm

In another desperate effort to destroy whatever tiny specks of credibility I might have in the scientific community, I’m going to post today about sea monsters. Yes, sea monsters, or, at least, one sea monster in particular. Most people know that various unidentified carcasses of mysterious creatures have been washed ashore over the years, and identified by lay-people as the remains of surviving prehistoric monsters. It’s true that in at least some cases these carcasses have later been identified as dead basking sharks, or as decomposing sperm whales, but there are an equal amount of other cases where no satisfactory identification has been proposed.
Of course that doesn’t automatically mean that the carcasses in question really did represent large, hitherto unknown marine animals (what we might technically term aquatic cryptids), but scientists should at least be interested enough in this possibility to look at the evidence and use their expertise in resolving these mysteries. There are two points worth making here. Firstly, it is well known (or at least I hope it is) that at least some large vertebrate species await discovery, especially marine ones (if you don’t believe me see: Paxton 1998, 2001, Raynal 2001, Solow & Smith 2005), and indeed this is constantly verified by the continuing recognition and description of new large marine vertebrates species (e.g. Mesoplodon perrini in 2002, Balaenoptera omurai in 2003, Orcaella heinsohni in 2005). Secondly, qualified scientists have looked, and do look, at the ‘sea monster’ literature, and it just isn’t fair or accurate to think that this subject isn’t worthy of good scientific attention.
A mid-length article published in 2001 included most of my thoughts on aquatic cryptids (Naish 2001). Some of the assertions made there are now outdated, or should be revised or corrected, but by and large I’m told that it’s a useful review. Unfortunately it seems that other researchers have either had trouble obtaining this article (the journal in which it was published appeared erratically, was mail-order only, and now appears impossible to obtain), or have remained completely unaware of it (and there’s the third possibility that they do have access to it, and are aware of it, but have just chosen to ignore it). While (I hope) I’m not arrogant enough to think that I hold the ultimate or most-useful opinions on this area, I do think that there are some comments and observations in the article that should be more widely acknowledged.
One of the most interesting ‘mystery carcass’ tales is that of ‘Gambo’*, a flippered sea creature apparently washed ashore on a Gambian beach in 1983. Most of the analysis and speculation about this carcass has come from prominent cryptozoologist Karl Shuker, and it’s no secret that Shuker has consistently interpreted Gambo and other cryptids as ‘prehistoric survivors’; viz, as late surviving representatives of groups otherwise thought to have gone extinct in the Mesozoic. I’ve disagreed with this on several occasions and indeed Shuker and I have been at loggerheads over what I term the ‘prehistoric survivor paradigm’ [go here for another post on this subject]. Regardless of how the reported carcass is interpreted, the significance of the case is that the exact location of Gambo’s burial site is known. Here’s where the story become topical. The new issue of Animals & Men arrived a few weeks ago, and therein is news that an official attempt to locate and exhume the carcass is to go ahead later this year (assuming, of course, that the carcass exists). Chris Moiser (of Plymouth College of Further Education) has led previous efforts to locate it while on fieldwork in the Gambia, but this time things will be done properly.
* Cryptozoologists have a really, really annoying habit of coming up with cutesy pet names for cryptids: Shuker is among them (the respective cryptozoologists, not the cutesy pet names) and coined ‘Gambo’ for use in his book In Search of Prehistoric Survivors.
So in the interests of disseminating my thoughts on Gambo to a wider audience, here is what I wrote about it in Naish (2001, pp. 88-89)…
‘Gambo’ - the Gambian sea serpent
Evidence comparable to the NHT is a carcass allegedly examined by Owen Burnham in June 1983 at Bungalow Beach, Gambia. First reported in a letter to BBC Wildlife, Burnham’s case has received publicity thanks to texts devoted to discussion of it by Bright (1989) and Shuker (1986a, b, 1993, 1995, 1996, 1998). Burnham claims he knows exactly where this is buried: no excavation there has yet been carried out. Owen Burnham was not the sole witness of the Gambian carcass: it is documented that his father, sister and brother observed it as well. That their accounts tally with his (though I understand that they reported somewhat differing measurements (G. Inglis pers. comm. 1997)) does support authenticity of the account. However, I find it very difficult to believe that, as a competent naturalist, Burnham failed to take any kind of sample whatsoever. Clearly, Burnham could not be expected to pickle the carcass, or hack through it to pull out a potentially diagnostic organ or bone, but why did he not think to retain one piece of skin, a flipper, a tooth? Furthermore, no sketches drawn from the carcass, or photographs, exist. Burnham’s several renditions of the creature were drawn on his return to the UK and not during his time in the Gambia. I bring attention to these points, not to attempt in any way to discredit Burnham or his report, but to illustrate the apocryphal and anecdotal nature of the account. As appealing as it is in potentially promising a bona fide sea serpent carcass, it is unfortunately no better than the many tales of lost sea serpent carcasses from the 18th and 19th centuries.
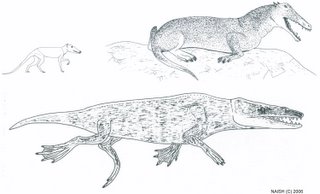
Burnham’s description is essentially that of a small, short-necked plesiosaur, and he is adamant that it was not a whale. The only cetacean that might conform in some features to the Gambian carcass is Tasmacetus shepherdi, the poorly known Shepherd’s or Tasman beaked whale, a species known from few specimens and only two probable sightings from the field (Watkins 1976, Laughlin 1996). Burnham discounted this species (and all other cetaceans) as the identity for the carcass and, though I feel based on Burnham’s descriptions that this identification is not satisfactory, I note that some Tasmacetus carcasses do bear more than a passing resemblance to Burnham’s drawings. Mead’s (1989) photographs of a beached Tasmacetus specimen from Punta Buenos Aires is superficially not unlike Burnham’s drawings (Fig. 8). In contrast to some artistic depictions of this species, these photos reveal an elongate, forceps-jawed rostrum, a poorly expressed melon, and prominent teeth along the lengths of the jaws. I also note some problems in Burnham’s narrative. For example, he notes that no mammary glands were present, and that any external genitalia were ‘too damaged to be recognisable’. However, the animal’s underside could not be examined so it is not clear how these observations could have been made. Preliminary excavations in the approximate area of ‘Gambo’s’ alleged burial have not been successful though, clearly, extensive excavations would be more satisfactory (Anon. 1997, Downes 1997).
The picture above is from…
http://upload.wikimedia.org/wikipedia/en/0/0b/Gambo.jpg
Yes, Gambo has its own Wikipedia entry.
Refs - -
Anon. 1997. In search of Gambo. Animals & Men 14, 11-13.
Bright, M. 1989. There are Giants in the Sea. Robson Books (London), pp. 224.
Downes, J. 1997. Mission Impossible: the search for ‘Gambo’. Uri Geller’s Encounters 9, 50-53.
Laughlin, C. 1996. Probable sighting of Tasmacetus shepherdi in the South Atlantic. Marine Mammal Science 12, 496-497.
Naish, D. 2001. Sea serpents, seals and coelacanths. In Simmons, I. & Quin, M. (eds) Fortean Studies Volume 7. John Brown Publishing (London), pp. 75-94.
Paxton, C. 1998. A cumulative species description curve for large open water marine animals. Journal of the Marine Biologists Association, U.K. 78, 1389-1391.
- . 2001. Predicting pelagic peculiarities: some thoughts on future discoveries in the open seas. In Heinselman, C. (ed) Dracontology Special Number 1: Being an Examination of Unknown Aquatic Animals. Craig Heinselman (Francestown, New Hampshire), pp. 60-65.
Raynal, M. 2001. Cryptocetology and mathematics: how many cetaceans remain to be discovered? In Heinselman, C. (ed) Dracontology Special Number 1: Being an Examination of Unknown Aquatic Animals. Craig Heinselman (Francestown, New Hampshire), pp. 75-90.
Shuker, K.P. N. 1986a. The Gambian sea-serpent. The Unknown, September 1986, 49-53.
- . 1986b. The Gambian sea-serpent. The Unknown, October 1986, 31-36.
- . 1993. Gambo – the beaked beast of Bungalow Beach. Fortean Times 67, 35-37.
- . 1995. In Search of Prehistoric Survivors. Blandford (London), pp. 192.
- . 1996. The Unexplained. Carlton Books (London), pp. 224.
- . 1998. Fishy tales. The X Factor 45, 1249.
Solow, A. R. & Smith, W. K. 2005. On estimating the number of species from the discovery record. Proceedings of the Royal Society B 272, 285-287.
Watkins, W. A. 1976. A probable sighting of a live Tasmacetus shepherdi in New Zealand waters. Journal of Mammalogy 57, 415.