This post follows on from the previous three (part I
here, part II
here, and part III
here), and if you read to the end of part III you’ll know that I got as far as talking about the media attention that the Naish
et al. (2004) paper received in late November 2004. As mentioned, I spoke to lots of journalists, and among them, one in particular had something very interesting to say. Unfortunately I forget his name, but I do recall that he was based on the Isle of Wight. His recollection was that the specimen had in fact been brought to the attention of the media before. Basing my conclusions on what happened with
Eotyrannus, this is almost certainly correct.
Steve Hutt’s plan with Isle of Wight dinosaur discoveries (and you’ll recall from the previous posts that Steve was the first person other than Gavin Leng to become acquainted with MIWG.7306) has always been to get publicity both on the discovery of a specimen,
and on the publication of the formal description. So
Eotyrannus was in the newspapers as a new dinosaur discovery in 1998, and then again in 2001 when it was formally named and described (Hutt
et al. 2001). Most of us hold off on talking to the press until our technical work has been published, but I’m not knocking Steve for his double-whammy approach, as the media are evidently interested enough to cover these stories twice.
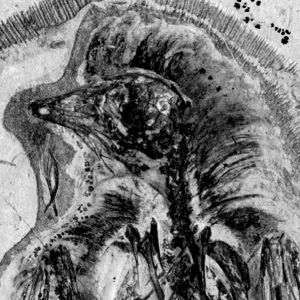
So while I’ve never seen the relevant articles, it seems that MIWG.7306 was reported in the newspapers at the time of its discovery. It also turns out that a semi-technical report was published on the specimen, and to my annoyance I didn’t find out about this until recently. The article in question is by Jon Radley, well known for his excellent work on Wealden stratigraphy, and it includes two paragraphs on the specimen and a photo (Radley 1997, pp. 108-109). The relevant section reads (though with some typos corrected)…
A brachiosaurid sauropod vertebra from the Wessex Formation (Wealden Group, Lower Cretaceous) of Sudmoor Point
In the autumn of 1993 Mr. G. Leng discovered a large sauropod vertebra derived from a plant debris bed exposed in the cliff top approximately 1 km northwest of Chilton Chine (SZ 399825). Mr. Leng has generously donated this important specimen to the Museum of IW Geology (MIWG 7306) with the permission of the National Trust. The specimen is preserved in a large, well-cemented sideritic concretion and is consequently only partly crushed. Small quantities of pyrite occur in the bone material and appear to be quite stable.
The bone is 0.75 m long and now ranks as the largest sauropod vertebra in the museum collection. Mr. S. Hutt has identified it as a cervical (neck) vertebra of an adult brachiosaurid sauropod. It is deeply socketed and possesses large prezygapophyses and postzygapophyses. The nature of construction is extremely light with well developed networks of pleurocoels (air chambers). From its size, one can calculate that it came from an animal approximately 22 to 25 m in length. Most brachiosaurid remains discovered so far on the Island are of considerably smaller animals – Radley & Hutt (1993) provided outline details of a recent find. It is hoped that the vertebra will be on temporary display in the museum in the near future.

A few things make Jon’s article particularly interesting. The ‘autumn of 2003’ date he provides is different from the 2002 one I was provided by MIWG staff, and the 750 mm length he provides is of course accurate (and presumably so because it’s the centrum length alone). The article also includes the first ever figure of the specimen, showing it in its unprepared, siderite-encased case (a scan of that figure is included here). Of course the article doesn’t in any way diminish the value of the final published description (Naish
et al. 2004) and is nothing more than an initial, preliminary report. I just wish that I’d known about it when writing
Dinosaurs of the Isle of Wight and the final MIWG.7306 paper, as then I could have cited it. Oh well.
It turns out that the final published description – while not bad as descriptions go – only really scratches the surface in terms of what information we can learn from MIWG.7306. Previously I discussed the incredible fact that brachiosaurid vertebrae are as much as 80 or 90% air, and that this degree of pneumatisation might mean an awful lot as goes physiology and biology. Because MIWG.7306 is broken into halves, making examination of its pneumatic interior possible, a current project is to get lots more data about pneumaticity out of it. That’s ongoing however, so I don’t want to talk more about it now.
MIWG.7306 also gives us new information on the diversity and distribution of brachiosaurids. MIWG.7306 clearly shares a number of detailed features only with
Brachiosaurus of the Late Jurassic of North America and eastern Africa, and with
Sauroposeidon of the Early Cretaceous of the USA, and as discussed in the paper (Naish
et al. 2004) it seems that MIWG.7306 might even be phylogenetically intermediate between these two forms. One of the big questions concerning brachiosaurids is which other sauropods are members of this clade too, and this is an area currently under study by my colleague Mike P. Taylor, who has a lot of new data (and some new species) on this subject.
But if MIWG.7306 represents an animal that is ‘phylogenetically intermediate’ between
Brachiosaurus and
Sauroposeidon, and given that it’s been publicised as a ‘new’ dinosaur, why didn’t we name it? That’s a good question, and there are two answers.
Firstly, most experts agree that specimens should only be named as new taxa if they can be can be shown to be
diagnostic: that is, they possess unique features which allow them to be differentiated from other taxa. Given that taxa evolve from ancestors, and evolve into descendants, there aren’t sharp boundaries between species and genera – rather, they grade into one another. Consequently, so-called diagnostic features must also, at some stage in any lineage, morph into the slightly different conditions present in ancestors and/or descendants, and multiple intermediate conditions must exist between any two ‘diagnostic’ end-states. We therefore, arbitrarily, chose cut-off points in how much variation we tolerate within any taxon – in other words, we arbitrarily decide how we chop up a lineage into those units we call species and genera. As a rough rule of thumb this all works, more or less, so long as it’s understood that species are artificial segments of lineages (though having said that, any species that can be shown to be the ‘end point’ of its respective lineage is a clade, and of course this applies to living species given that they don’t have descendants). Anyway, I’m going off here at a tangent: this is the sort of heavy philosophical stuff that systematists have been arguing over for decades.
MIWG.7306 possesses loads of anatomical features that are also present in
Brachiosaurus and
Sauroposeidon, and it even shares some features that are present in
Sauroposeidon and not in
Brachiosaurus but, so far as we can tell, it doesn’t possess any features that are unique and thereby allow it to be reliably differentiated from all other sauropods. That’s almost certainly is an artefact resulting from the fact that we only have two cervical vertebrae of course – if we had the whole skeleton things would be different. But, as it stands, MIWG.7306 cannot presently be diagnosed as a new species.

Actually, for a while I’ll admit that we thought that MIWG.7306
could be diagnosed. One of several unsolved mysteries about the specimen is the identity of a bizarre, oddly shaped chunk of bone found encased in the same nodule (Dave Martill is holding it in the adjacent image). Smoothly convex on one side, but with a series of subparallel ribs on the other, it was tentatively identified by David Cooper as part of the apex of the neural spine. I thought later on that it might be a partial diapophysis*, as those of brachiosaurids can descend ventrally from the side of the centrum as plate-like processes with smoothly convex lateral surfaces. If this identification is right, then the diapophysis of MIWG.7306 is uniquely odd, as it possesses a small rhomboidal opening near its (presumed) posterior border. Such a feature is unknown elsewhere in sauropods, and it’s the sort of feature we might chose to regard as diagnostic. I now have serious doubts about my identification of this object as a diapophysis, and I think that David was right with the neural spine identification. Images of the object were shown to sauropod-obsessed colleagues, and they remains uncertain as to what it is however, so the thing remains mysterious.
* In tetrapods with two-headed ribs, the more ventral rib head contacts a facet on the vertebra known as the parapophysis (plural parapophyses), while the more dorsal rib head contacts a facet known as the diapophysis (plural diapophyses). The positions of the parapophyses and diapophyses change along the length of the vertebral column, and indeed their relative positions allow us to identify where in the sequence an isolated vertebrae came from.
The second answer [to the question: why didn’t we name it?] is that MIWG.7306 comes from a geological unit (the Wessex Formation) where there are already lots of named sauropods, virtually all of which are based on non-overlapping fragments, such as vertebrae. What’s more, some of these (notably
Eucamerotus, a form named for dorsal vertebrae), seem to be good honest brachiosaurids closely related to
Brachiosaurus (and hence to MIWG.7306). It’s possible and perhaps likely that some of the other Wessex Formation sauropods,
Eucamerotus among them, actually represent the same taxon as MIWG.7306, though of course this can’t be tested until we have good, more complete specimens (here you’ll recall the Barnes High brachiosaurid, still floating in scientific limbo). In view of this situation it would be regarded as bad practise to coin a new name for MIWG.7306.
Partly because it’s easier to say than ‘MIWG.7306’ we’ve elected to use a totally unofficial nickname for the taxon represented by MIWG.7306, and this is ‘Angloposeidon’, coined by brachiosaurophile Mike P. Taylor (who is irritated by my continual reference to him as such*). So long as it stays on the internet we’re ok: it must NOT be published!
* So why do I do it? Because there is already a well known Mike Taylor in the world of tetrapod zoology: the marine reptile expert Mike A. Taylor.

What do other experts think of MIWG.7306? During the long period of time in which the manuscript was in preparation I spoke to several European colleagues who told me of new sauropods from Portugal and Spain that would easily outclass MIWG.7306 in terms of size. I had this on my mind all the way through the submission process, and at any time I expected there to be some report of a new European sauropod that had a total length exceeding 30 m. But even today such discoveries have yet to materialise, and having now seen some of the specimens in question I know that they fail to come close to the 20 m + estimated for MIWG.7306. The accompanying image – a composite produced by Matt Wedel and Mike – shows the forelimb of one of these ‘gigantic’ European sauropods (with me for scale) adjacent to the Chicago mount of
Brachiosaurus (with Mike for scale). ‘Angloposeidon’ was about equivalent in size to
Brachiosaurus so far as we know, and as you can see,
Brachiosaurus knocks spots off the European animal (which, by the way, is as yet undescribed and unnamed).
The MIWG.7306 paper hasn’t been cited much in the literature, but this results from the fact that bugger all has been published on Lower Cretaceous British sauropods since 2004. Matt Wedel – pneumaticity and giant brachiosaurid expert – has certainly been interested and even came to see the specimen in March 2004. He’s agreed with our interpretations, and in recent publications on
Sauroposeidon has noted that MIWG.7306 shows that Britain was once home to a close relative of this Oklahoman giant (Wedel 2005: free pdf
here).
Sauroposeidon is from the Aptian-Albian Antlers Formation, whereas the Wessex Formation that yields MIWG.7306 is just a little older, being late Barremian in age. We also know that similar giant brachiosaurids were present in the Aptian-Albian Cloverley Formation of Montana, as evidenced by a single cervical vertebra held today at the Yale Peabody Museum. The specimen is from a juvenile brachiosaurid but, as Wedel (2005) noted, at 47 cm in length the specimen ‘is longer than the vertebrae of many adult sauropods’ (p. 55). Footprints produced by
Sauroposeidon, or by a similar, closely related brachiosaurid are known from the Glen Rose Limestone of the Paluxy River, Texas. At up a metre in diameter, they must have been produced by a real giant, and, among roughly contemporaneous North American sauropods, only
Sauroposeidon is big enough (Wedel 2005).
And that’s pretty much the whole story up to now. The main message I suppose you should take away is that producing a technical paper on a specimen – even a short one devoted to the description of a single bone – can be an absurdly drawn-out, lengthy affair literally years in the making, and this is all the more so when other projects and life in general get in the way. Discovered by an enthusiastic amateur who donated the specimen to his local museum (Gavin Leng), prepared by an amateur with scientific training (David Cooper), and eventually described technically by a team of palaeontologists, the MIWG.7306 story is also a nice example of the sort of successful collaboration that can result if people work together.
However, there is also a sad ending to this story. David Cooper, who devoted so much time to the specimen and initiated the research that culminated in the paper, had been suffering for some time from cancer, and by 2005 he knew his condition was terminal. Late in June 2005 I received an unexpected and saddening phonecall. Given that I received this news the day before his funeral, I was unable at such short notice to make the arrangements to attend. This, I regret.
For the latest news on Tetrapod Zoology please go
here.
Refs - -
Hutt, S., Naish, D., Martill, D. M., Barker, M. J. & Newbery, P. 2001. A preliminary account of a new tyrannosauroid theropod from the Wessex Formation (Early Cretaceous) of southern England.
Cretaceous Research 22, 227-242.
Naish, D., Martill, D. M., Cooper, D. & Stevens, K. A. 2004. Europe’s largest dinosaur? A giant brachiosaurid cervical vertebra from the Wessex Formation (Early Cretaceous) of southern England.
Cretaceous Research 25, 787-795.
Radley, J. 1997. Geological report 1993-1994.
Proceedings of the Isle of Wight Natural History and Archaeology Society 13, 107-114.
Wedel, M. J. & Cifelli, R.L. 2005.
Sauroposeidon: Oklahoma’s native giant.
Oklahoma Geology Notes 65, 40-57.
Labels: archosaurs, dinosaurs, Mesozoic, sauropods
 Following the metriorhynchid post a few people have asked for more details on the Oxford Clay fauna. Luckily the accompanying image shows you all you need to know :) [for the same image at much better resolution visit the relevant page at my flickr site]. Note that it’s a CARTOON (in fact it was deliberately made to go on the front of a t-shirt). The term Oxford Clay refers to a series of mudrocks, formally grouped together as the Oxford Clay Formation, laid down during the Callovian and early Oxfordian of the Middle Jurassic, a time when shallow tropical seas covered much of Europe. Layers of Oxford Clay several hundred metres thick were deposited over Britain and northern France. Judging by the huge number of fossils discovered in the unit, organic productivity was extremely high and rich sources of terrestrially-derived nutrients must have been discharged into the sea. Pretty much everything you’d want to know about the Oxford Clay Formation is included in Martill & Hudson (1991).
Following the metriorhynchid post a few people have asked for more details on the Oxford Clay fauna. Luckily the accompanying image shows you all you need to know :) [for the same image at much better resolution visit the relevant page at my flickr site]. Note that it’s a CARTOON (in fact it was deliberately made to go on the front of a t-shirt). The term Oxford Clay refers to a series of mudrocks, formally grouped together as the Oxford Clay Formation, laid down during the Callovian and early Oxfordian of the Middle Jurassic, a time when shallow tropical seas covered much of Europe. Layers of Oxford Clay several hundred metres thick were deposited over Britain and northern France. Judging by the huge number of fossils discovered in the unit, organic productivity was extremely high and rich sources of terrestrially-derived nutrients must have been discharged into the sea. Pretty much everything you’d want to know about the Oxford Clay Formation is included in Martill & Hudson (1991). A smallish, gracile-snouted pliosaurid, Peloneustes, is shown at far left in the scene. There are some indications that Peloneustes had particularly big wing-like paddles (Bakker 1993), but further study is needed to confirm this. Its slim snout suggests that it wasn’t a macropredator. The small, short-necked plesiosaur near the sea floor is the pachyostotic pliosaurid Pachycostasaurus, described by Cruickshank et al. (1996). Only known from one juvenile specimen that would have been about 3 m long, it’s poorly known but seems to have been a specialised bottom-cruising form. It might then have preyed on benthic animals, such as burrowing shrimps. The adjacent image of a Peloneustes skeleton is borrowed from Palaeos.com.
A smallish, gracile-snouted pliosaurid, Peloneustes, is shown at far left in the scene. There are some indications that Peloneustes had particularly big wing-like paddles (Bakker 1993), but further study is needed to confirm this. Its slim snout suggests that it wasn’t a macropredator. The small, short-necked plesiosaur near the sea floor is the pachyostotic pliosaurid Pachycostasaurus, described by Cruickshank et al. (1996). Only known from one juvenile specimen that would have been about 3 m long, it’s poorly known but seems to have been a specialised bottom-cruising form. It might then have preyed on benthic animals, such as burrowing shrimps. The adjacent image of a Peloneustes skeleton is borrowed from Palaeos.com. Finally, ichthyosaurs. Only thunniform Ophthalmosaurus – named for its immense eyes – is known from the Oxford Clay, though jaw fragments sporting big teeth have been suggested to belong to a second genus by some workers. However, these fragments might belong to Ophthalmosaurus as, while usually characterised as toothless, there are now indications that this wasn’t so. The huge eyes suggest that Ophthalmosaurus was a deep-diver, perhaps hunting well offshore for deep-sea cephalopods. The adjacent image is taken from the Saurier Museum Rundgang site.
Finally, ichthyosaurs. Only thunniform Ophthalmosaurus – named for its immense eyes – is known from the Oxford Clay, though jaw fragments sporting big teeth have been suggested to belong to a second genus by some workers. However, these fragments might belong to Ophthalmosaurus as, while usually characterised as toothless, there are now indications that this wasn’t so. The huge eyes suggest that Ophthalmosaurus was a deep-diver, perhaps hunting well offshore for deep-sea cephalopods. The adjacent image is taken from the Saurier Museum Rundgang site.